Elemental Imaging of Biological Samples Using J200 Tandem LA – LIBS
by Dr. Alexander Bolshakov
Introduction
Laser Ablation ‒ Inductively Coupled Plasma ‒ Mass Spectrometry (LA-ICP-MS) is broadly used today as a powerful microanalytical technique for spatially-resolved trace elemental and isotopic analysis of biological samples. However generally it is not suitable for analyzing major elements. Strong isobaric interference and elevated background cause difficulties in measuring nonmetals by LA-ICP-MS, especially carbon, hydrogen, oxygen, and nitrogen that are major components of biological tissues. On the contrary, these elements are easily measured by Laser Induced Breakdown Spectroscopy (LIBS). Evidently, optical and mass spectra ought to be acquired simultaneously from every laser ablation pulse. Applied Spectra’s J200 Tandem LA‒LIBS instrument enables such measurements of all the elements in a sample by combining the analytical merits of both techniques LA-ICP-MS and LIBS.
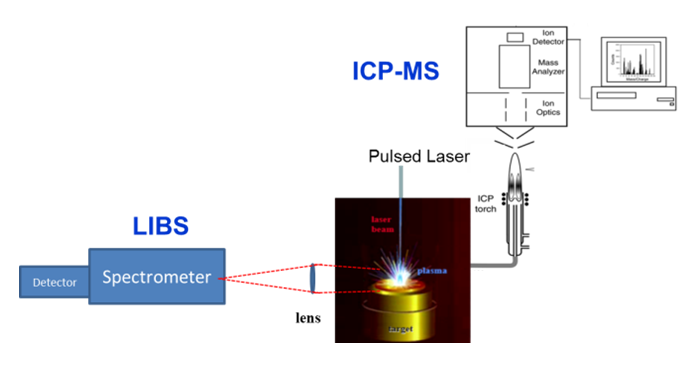
Representation of laser ablation as the direct solid sampling process with subsequent analysis of photons from the induced plasma using laser induced breakdown spectroscopy (LIBS) and by transporting the ablated mass to an inductively coupled plasma (ICP) with mass spectrometry (MS).
A laser beam can be tightly focused into a spot of several microns in diameter. Accordingly, Tandem LA‒LIBS measurements provide high imaging definition with very high sensitivity for the trace elements at each spatial location where laser ablation pulses are directed. Tandem LA‒LIBS can perform 2D (or 3D, layer-by-layer in depth) chemical maps that represent quantified elemental distributions in biological samples.
Elemental imaging at the cellular range becomes possible in correlation or normalization on density and composition of the organic matrix. It is especially important for pathological tumors subjected to investigation of efficiency of anti-cancer drugs. High spatial resolution of chemical imaging is required because cancer cells are unevenly distributed forming heterogeneous clusters of different shapes and sizes.
Other techniques currently used for fine element-specific bioimaging are based on X-rays (μXRF, μPIXE, SEM-EDX, XFM) or highly sophisticated mass spectrometry (nano-SIMS, FT-TIMS). The former require either synchrotron radiation or radioactive or proton sources. The later require placing a sample into a vacuum chamber. While those approaches can offer submicron resolution, the Tandem LA‒LIBS has broader elemental and isotopic coverage, wider spatial range, operates simpler and faster, and is well suited for quantitative imaging of tissues, whole organs and organ structures, metabolism processes, growth patterns, nanoparticle uptake, transport, protein and single cell analysis. Tandem LA‒LIBS may involve only minimal or no sample preparation.

Applied Spectra’s J200 LA-LIBS Instrument
Several examples of using the J200 Tandem LA‒LIBS Instrument for bioimaging are following.
Imaging of human pleural mesothelioma
The J200 Tandem LA‒LIBS instrument equipped with nanosecond-pulsed Nd:YAG laser operating at forth harmonic (266 nm) was used for mapping a human tumor slice from an individual previously treated with cisplatin as anti-cancer drug. The J200 Tandem instrument included a broadband 6-channel optical spectrometer (covering 190‒1040 nm) fitted with multi-channel CCD detectors. ICP-MS analysis was performed by a Thermo iCAP Q instrument. The tumor section was scanned by a focused laser beam in a line scan pattern using distances between lines equal to the size of the ablation crater (Ø40 μm). Lateral resolution of 40 μm was sufficient for medical interpretation of the results but further increasing the resolution would unnecessarily prolong total measuring time.1 The obtained distribution images for selected elements are shown in Figure 1.
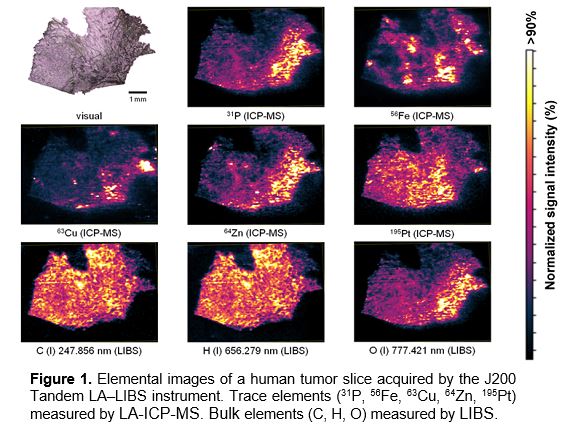
The Tandem LA‒LIBS measurements resulted in images of trace elements (Pt, P, Fe, Zn, Cu) acquired by LA-ICP-MS along with abundant elements (C, H, O, Na, K, Ca, Mg) acquired by LIBS at the same time. Carbon and hydrogen were distributed fairly homogeneously over the bulk of the tissue. Some variations in C and H spectral intensities are probably indicative of different tissue density at respective locations. A correlation coefficient for C and H signals was 0.96 revealing a constant C/H ratio throughout the tissue. Phosphorus and oxygen were unevenly distributed but well correlated with tumor regions and to each other. This can be explained by elevated phosphates, phospho-lipids, DNA and activated proteins (phosphorylated amino acids) in the tumor regions.
Zn and Cu exhibited similar distributions acting as known cofactors of proteins. Iron concentration was variable and unrelated to other distributions.
Platinum delivered by cisplatin accumulated mostly in healthy tissue. A resistance mechanism might occur hampering the drug uptake and requiring more time to enter the tumor region.
Na and K were rather homogeneously distributed (not shown in Fig. 1) with good correlation to each other. These two elements have similar biological functions to maintain membrane pressure in cells. Mg and Ca also play similar physiological roles, and therefore they demonstrated distributions similar to each other (not shown in Fig. 1).
A further study confirmed a perfect correlation between the sodium images obtained by LA-ICP-MS at the mass 23Na and by LIBS at the 589-nm Na optical emission line. These two sodium distributions were numerically compared yielding a Pearson correlation coefficient R = 0.918, which demonstrated excellent reliability of the results.2
Imaging of porcine whole skin in vitro
Silver nanoparticles are widely used in cosmetics and skin care products. A possibility of Ag penetration through damaged or abraded skin (inflammation, wounds, excessive sunlight, and regular shaving) raises reasonable questions about consumer safety. The J200 Tandem LA-LIBS instrument with nanosecond solid-state Nd:YAG laser operating at wavelength 266 nm was used to map the top layers of the epidermis from isolated pig skin samples.3 In this study ICP-MS analysis was performed by a Bruker Aurora Elite instrument.
After exposure to silver nanoparticles, the skin samples were embedded in paraffin and scanned by a laser beam (Ø35 μm) in a linescan pattern using distances between lines of 70 μm. The skin samples were exposed to citrate stabilized Ag nanoparticles (45 nm) at concentrations 47, 470 and 750 μg/ml, and also mixed with other popular cosmetics ingredients. Laser pulse repetition rate was 10 Hz. The quantified elemental images for Ag, C, P, and S are provided in Figure 2. Carbon signal from paraffin was subtracted.
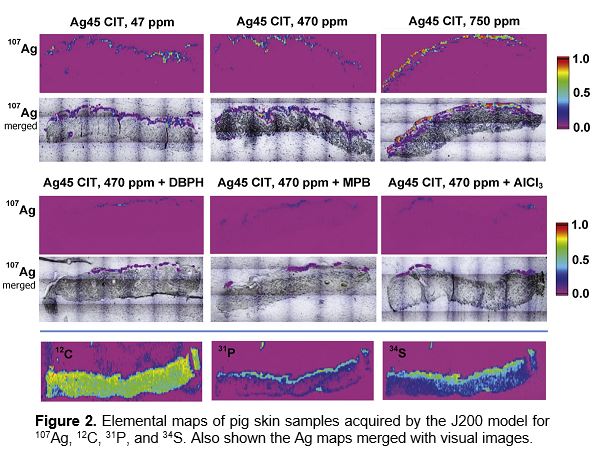
Strong Ag signals were detected at the corneal layer of epidermis and in the hair follicles. No clear trends of silver absorption in skin was found between concentrations and sizes of the applied Ag nanoparticles.
However, lower Ag penetration was observed for bigger Ag nanoparticles if blended with the chemicals used in cosmetics formulae.
The highest amount of Ag penetrated into skin was 0.45 ng/cm2. as measured by LA-ICP-MS using J200 Tandem LA-LIBS Instrument.
Imaging of rat brain in vitro
Neurological functions involve biochemistry of metals in brain, and their distributions reflect details of the brain architecture. An example of using J200 Tandem LA-LIBS Instrument model for elemental mapping of rat brain hemispheres is illustrated in Figure 3. The rates were treated with Mn administered either an nanoparticles or MnCl2 solution, which is often used as a contrast agent and marker for magnetic resonance imaging.
Copper and zinc are two disease-relevant metals within the brain. Mn, Cu, and Zn were measured using a protocol similar to the porcine whole skin analysis (see above). The results demonstrated that Mn nanoparticles tended to accumulate and remained longer in rat brains than MnCl2 solution.
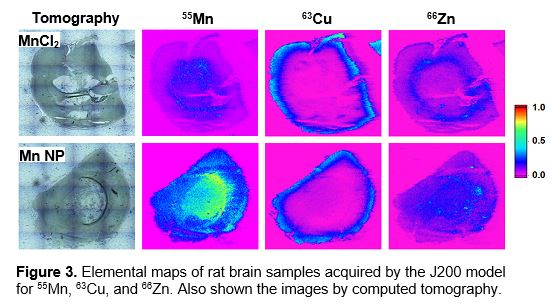
Zinc is an essential trace element for brain function. Zinc may inhibit infection due to Zn-containing cytokines secreted by the brain cells.
High dosages of copper may be toxic owing to its ability to change oxidation states in redox reactions.
Mapping of contaminated leaves
Detailed elemental mapping of vegetative organs can be used to determine pathways of uptake of available nutrient content, toxic elements from environmental pollution, effects of climate change and life functions of the plants. The J200 Tandem LA‒LIBS Instrument with nanosecond solid state Nd:YAG
laser operating at 266 nm was used to map plant leaves exposed to a pollutant solution. ICP-MS analysis was performed by an Analytik Jena PlasmaQuant Elite instrument. The leaves were scanned using a 150 μm laser spot size. The resulting elemental images are shown in Figure 4.
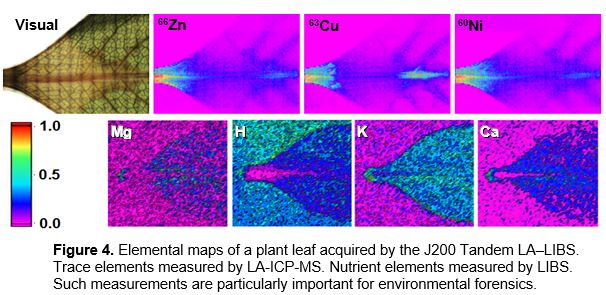
The pollutants (Zn, Cu, Ni) were mainly detected in midrib, vein and capillaries. The major nutrients (Mg, H, K, Ca) were distributed relatively uniformly over a leaf structure. Sensitivity of LIBS for Ca is great but can be low in ICP-MS, since the main isotope 40Ca is masked by 40Ar in ICP operation.
Femtosecond LA-ICP-MS and LIBS imaging
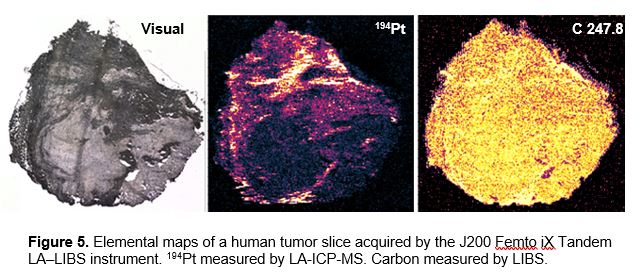
The J200 Femto iX Tandem LA‒LIBS instrument with a femtosecond laser source based on ytterbium diode pump technology operating at wavelength 343 nm was used for mapping a human lung tumor sample from a patient treated with cisplatin.
Femtosecond lasers are ideal for high-definition chemical imaging as they can create order of magnitude smaller and more regularly shaped ablation crates than nanosecond lasers do. Thus, spatial resolution in imaging is greatly improved.
Also, femtosecond lasers depose less thermal load on the sample and supress nonlinear matrix effects. This results in relaxed requirements for matrix-matched standard calibration. Femtosecond laser ablation generates smaller particles that are more efficiently transported into ICP-MS and more completely ionized by ICP. All these benefits lead to higher sensitivity, enhanced accuracy and precision of the chemical analysis as compared to nanosecond laser ablation.
Figure 5 demonstrates the results of mapping a tumor sample using the J200 Femto iX Tandem Instrument. Platinum (194Pt) was measured by ICP-MS at trace levels <20 µg/g, while carbon was measured by LIBS at concentration >25 wt%. Laser beam diameter was 10 µm. The elemental image resolution was significantly improved in comparison with the images shown in Figure 1.
The J200 Femto iX Tandem LA‒LIBS instrument is compact and very powerful at the same time.
References
- M. Bonta et al., J. Anal. At. Spectrom., 31, 252-258 (2016).
- M. Bonta et al., Spectroscopy, 32, 42-46 (2017).
- K. Domeradzka-Gajda et al., Toxicology Lett., 272, 38-48 (2017).

